Surgical Treatments: Artificial Urinary Sphincter (AUS)3
Depending on the severity of incontinence, an artificial urinary sphincter might be considered a long term solution. Talk to your doctor or urologist to explore whether this option is right for you.
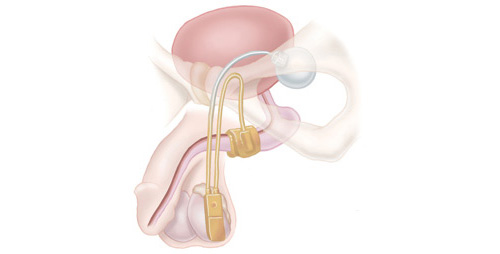
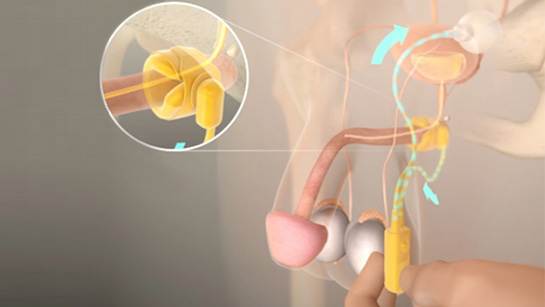
The artificial urinary sphincter (AUS) is a surgical device designed to mimic the same role as a healthy urinary sphincter, keeping the neck of the bladder closed until you wish to urinate. It works by automatically moving saline through three main components: an inflatable cuff, a pump and a pressurised balloon. These elements are connected by flexible tubing. The inflatable cuff fits around the urethra, the control pump is placed in the scrotum, and the pressurised balloon is implanted in the abdomen.
To urinate, you press and release the pump located in the scrotum several times to empty the cuff, allowing urine to flow out from the bladder. A few minutes after urination, the cuff automatically refills with saline. When the cuff is full, it once again compresses the urethra, holding it closed, providing continence.
What is an artificial urinary sphincter and how does it work?
Benefits:
- Designed to treat male stress urinary incontinence (SUI) following prostate surgery3
- Mimics a healthy sphincter, allowing you to urinate when desired3
- Offers most men with a weakened sphincter muscle the ability to achieve continence4
- Placed entirely inside the body, it is undetectable to others
- Requires good cognitive ability and manual dexterity3
Risks and Side Effects:3
As with any medical procedure, complications can occur. Operation of the device requires manual dexterity; some risks include, but are not limited to: device malfunction or failure which may require revision surgery, wearing away/loss of tissue (device/tissue erosion), inability to urinate (urinary retention), infection and postoperative pain.
Take the Quiz
Take our quiz to identify if you are experiencing urinary incontinence symptoms and help kick-start the conversation with your doctor.
Questions to Ask
Helpful hints on how to start the conversation with your GP or urologist, and other useful resources.
References
- Medline Plus. (n.d.). Retrieved December 13, 2019, from https://medlineplus.gov/ency/article/007373.htm.
- National Association for Continence. (n.d.). Retrieved December 13, 2019, from https://www.nafc.org/male-products.
- AMS 800™ Urinary Control System Operating Room Manual. American Medical Systems, Inc. 2011
- Poole K, Kerlin M, Wynne R. Prevalence and characteristics of urinary incontinence in a cohort of patients with chronic heart failure. Heart Lung. 2017; 46 (2): 67-73. https://www.heartandlung.org/article/S0147-9563(16)30224-2/fulltext
- Montague D. K. (2012). Artificial urinary sphincter: long-term results and patient satisfaction. Advances in urology, 2012, 835290. doi:10.1155/2012/835290
- Welk BK. Herschorn S. The male sling for post-prostatectomy urinary incontinence: a review of contemporary sling designs and outcomes. Br J Utol Int. 2011;109:328–344.
- AdVance™ Male Sling System Instructions for Use. American Medical Systems, Inc. 2010.
- Rehder P, Haab F, Cornu JN, et al. Treatment of post-prostatectomy male urinary incontinence with the transobturator retroluminal repositioning sling suspension: 3-year follow up. Eur Urol. 2012 Jul;62(1):140-5.
- Sturm RM, Guralnick ML, Stone AR, et al. Comparison of clinical outcomes between “ideal” and “nonideal” transobturator male sling patients for treatment of post prostatectomy incontinence. Urol. 2014 May;83(5):1186-8.
Results from case studies are not necessarily predictive of results in other cases. Results in other cases may vary. All images are the property of Boston Scientific. All trademarks are the property of their respective owners.
Disclaimer: Individual symptoms, situations, circumstances and results may vary. This information is not intended to be used for medical diagnosis or treatment or as a substitute for professional medical advice. Please consult your doctor or qualified healthcare provider regarding your condition and appropriate medical treatment.
These Videos are meant for informational purposes only and may not be indicative of clinical outcome. The opinions, procedures and patient care policies expressed or depicted in the videos are those of the physician or practice nurse and do not necessarily reflect the opinions, policies or recommendations of Boston Scientific Corporation or any of its employees.
This site is intended for Australian residents only. Please review the Boston Scientific Privacy Policy, for practices on the collection, storage, use and disclosure of your personal information.
CAUTION : Indications, contraindications, warnings and instructions for use can be found in the product labelling supplied with each device.